Marsden was to sit in the darkened room, wait for his eyes to adjust to the darkness, and then patiently stare at the screen, expecting to see nothing at all. Any scattered particles would hit a screen coated with zinc sulfide, which scintillates when hit with charged particles. The experiment involved firing alpha particles from a radioactive source at a thin gold foil. He later wrote that he felt it was a sort of test of his experimental skills. Marsden was not expected to find anything, but nonetheless he dutifully and carefully carried out the experiment. Rutherford, who didn’t want to neglect any angle of an experiment, no matter how unpromising, suggested Marsden look to see if any alpha particles actually scattered backwards. Rutherford had already been studying the scattering of alpha particles off a gold target, carefully measuring the small forward angles through which most of the particles scattered. In 1909, he and his colleague Hans Geiger were looking for a research project for a student, Ernest Marsden. In 1907 Rutherford returned to England, to the University of Manchester. (Rutherford is widely quoted as having said, “All science is either physics or stamp collecting”) The discovery earned Rutherford the 1908 Nobel Prize in Chemistry, which irritated him somewhat because he considered himself a physicist, not a chemist. In 1901 Rutherford and chemist Frederick Soddy found that one radioactive element can decay into another. There, he identified alpha and beta radiation as two separate types of radiation, and studied some of their properties, though he didn’t know that alphas were helium nuclei. Rutherford’s talent was quickly recognized, and in 1898 he took a professorship at McGill University in Montreal. 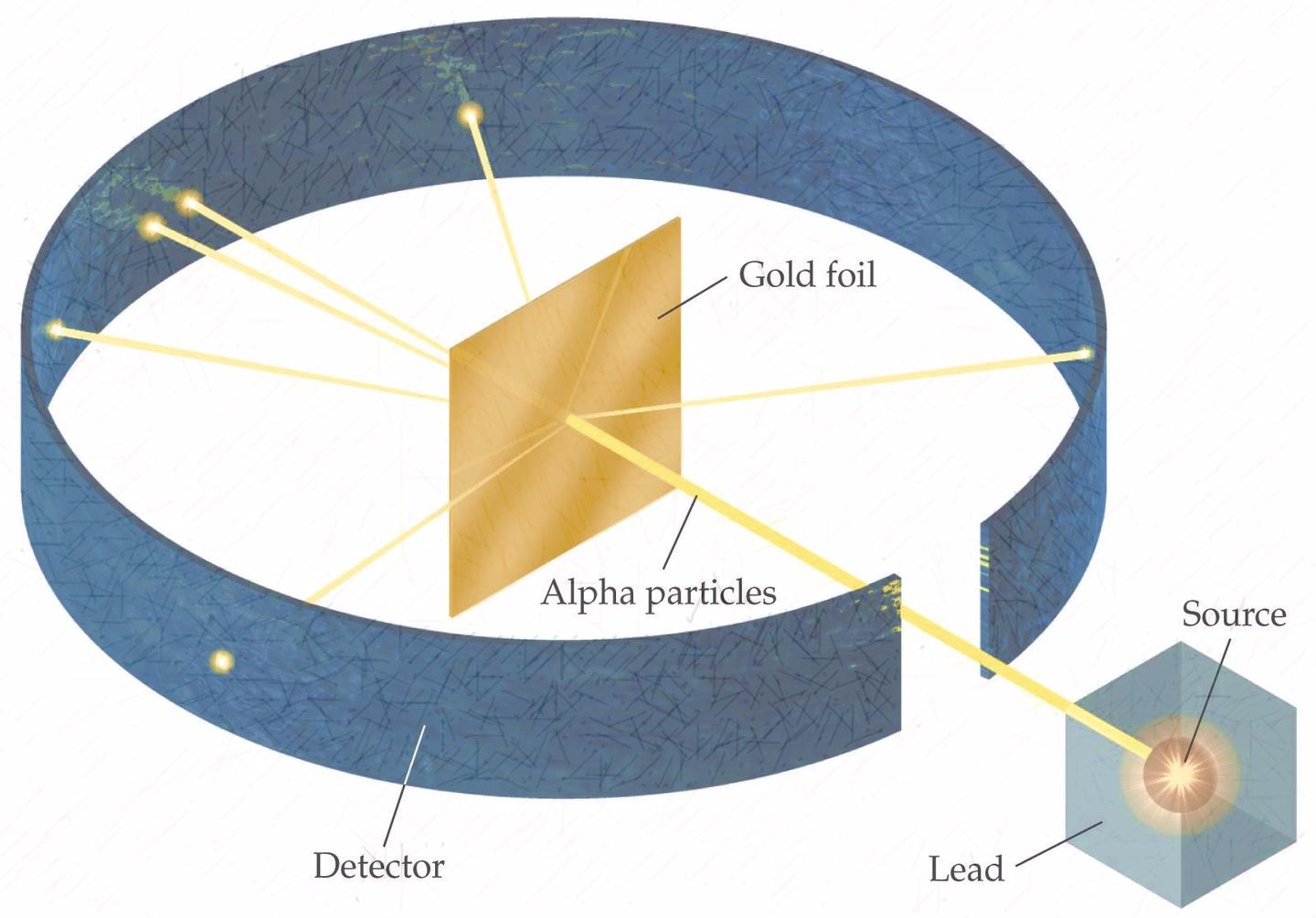
Upon receiving the news of this scholarship, Rutherford is reported to have said, “That’s the last potato I’ll ever dig.”Īt Cambridge, the young Rutherford worked in the Cavendish lab with J.J. 
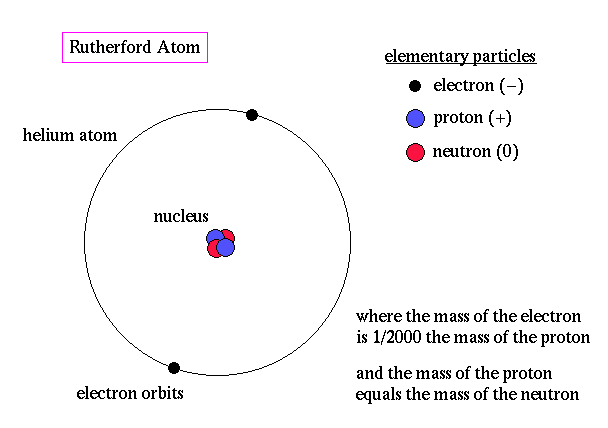
After college he won a scholarship in 1894 to become a research student at Cambridge. Growing up, he often helped out on the family farm, but he was a good student, and received a scholarship to attend the University of New Zealand. Rutherford’s explanation, which he published in May 1911, was that the scattering was caused by a hard, dense core at the center of the atom–the nucleus.Įrnest Rutherford was born in New Zealand, in 1871, one of 12 children. In the now well-known experiment, alpha particles were observed to scatter backwards from a gold foil. Rutherford called this news the most incredible event of his life. In 1909, Ernest Rutherford’s student reported some unexpected results from an experiment Rutherford had assigned him. May, 1911: Rutherford and the Discovery of the Atomic Nucleus 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
